Single-Use Acetabular Reamer - INSTRUCTIONS FOR USE
USE
The Single-Use Acetabular Reamer is intended for shaping the acetabular cavity in hip replacement surgery for use by trained orthopedic surgeons experienced in hip arthroplasty procedures.
Prior to using the reamer, the surgeon shall have given careful consideration to all aspects of the surgical intervention as well as to the limits of the reamer.
Technique for Surgical Use:
- Remove the reamer from the sterile package by opening and inverting the inner blister.
- Grasp the reamer at the edge of the molded baseplate and push onto the reamer shaft while pulling back on the locking sleeve, making a quarter turn to lock the reamer in place.
- Insert the reamer, in proper orientation, into the bony acetabulum and activate the surgical drill, advancing the reamer to the correct depth.
- Remove the reamer from the acetabulum and insert the properly sized prosthetic acetabular component.
- Remove the reamer from the reamer shaft by grasping the edge of the base plate, pulling back on the locking sleeve and rotating a quarter turn and pulling the reamer off the reamer shaft.
- Dispose of the reamer after a single use.
- During use should the reamer seize within the acetabulum, removing the reamer must be done by putting the power reamer in reverse or by removing the power reamer from the shaft and replacing it with a T- Handle instrument. Applying a counterclockwise rotation to the shaft will remove the reamer. Failure to follow this procedure can result in damage to the reamer and injury to the patient.
o Do not apply excessive force.
o Inspect device if seizure occurs.
o Do not reuse if structural damage suspected. - Adequate retraction of soft tissue during use is required to avoid possible damage to a functioning reamer from contact with a retractor.
REUSE
DO NOT REUSE OR REPROCESS. The Single-Use Acetabular Reamer is for use in a single procedure. A second procedure use is prohibited and may reduce the performance of the instrument and put the patient and the surgeon at risk. The reamer must be properly disposed of after each procedure.
EXAMINATION PRIOR TO USE
Single-Use Acetabular Reamer (sold sterile):
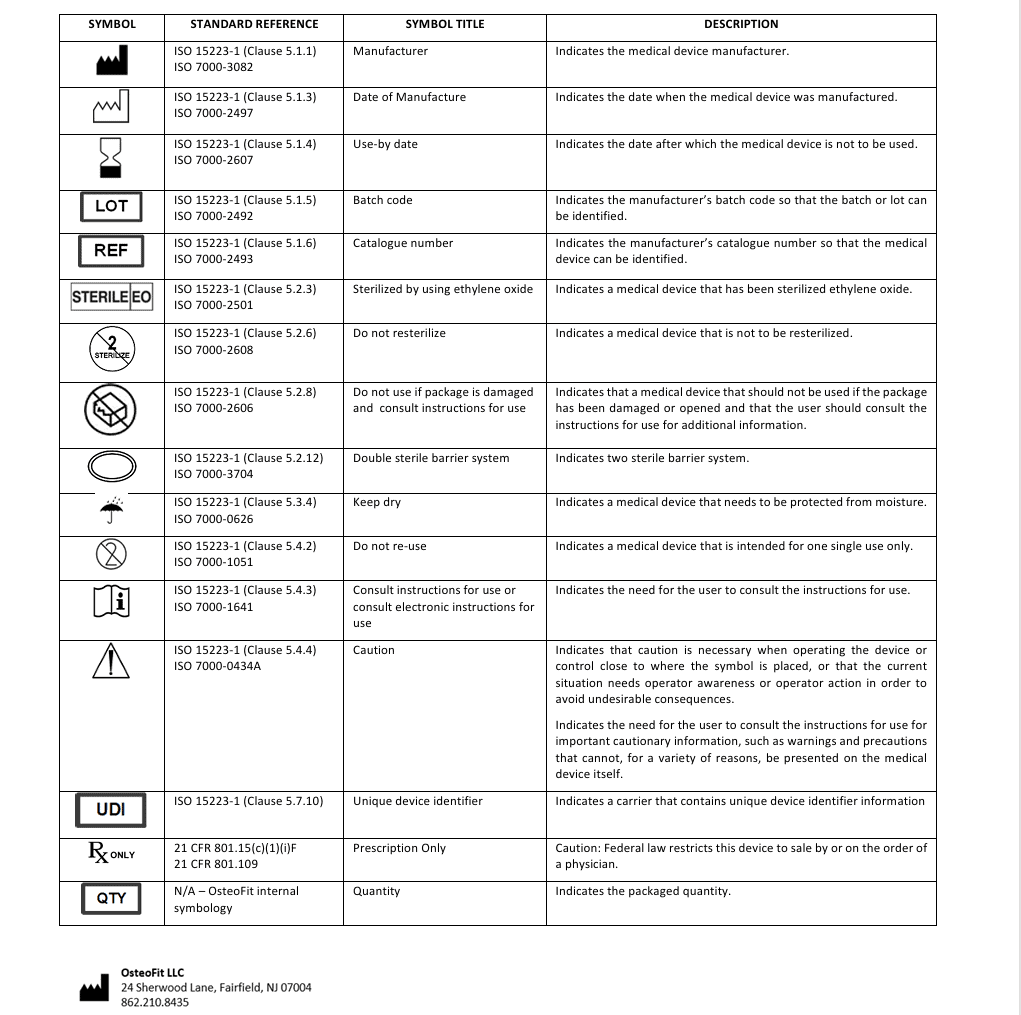
- Before utilization, it is necessary to verify the sterility expiration date, which is indicated as the “use by” date. Use after the expiration date may compromise sterility and device performance.
- It is recommended to verify the integrity of the instrument and original package before use. Sterility is ensured only if there is no trace of damage to the packaging.
- In case of damage to the packaging, or after opening of the packaging, re- sterilization of the instrument is strictly forbidden, regardless of the method that might be employed.
- In case of damage to the packaging, or after opening of the packaging, re- sterilization of the instrument is strictly forbidden, regardless of the method that might be employed.
PACKAGING
The Single-Use Acetabular Reamer is sold sterile and will be clearly labelled as sterile on the product label. The sterilization method applied is indicated on the product label. The sterile instrument packaging must be intact at the time of receipt and the integrity of the packaging shall be checked prior to use. Do not use if package is damaged. It is a double sterile barrier validated per ISO 11607.
MATERIAL INFORMATION
The Single-Use Acetabular Reamer is manufactured from Stainless Steel and Polycarbonate. Materials have been evaluated for biocompatibility in accordance with ISO 10993.
STERILIZATION
The Single-Use Acetabular Reamer is supplied by the company in a sterile condition. The instrument has been sterilized by Ethylene Oxide Gas Sterilization (noted as STERILE-EO on the label).
For shelf-life expiration, refer to the date printed on the product label. Meticulously inspect the package before use. Do not use the instrument if the package has been opened, is torn, or punctured, or if the seal has been broken.
Steam sterilization, including flash sterilization of the Single-Use Acetabular Reamer is NOT recommended.
TREATMENT POPULATION
- Osteoarthritis of the hip
- Rheumatoid arthritis of the hip
- Avascular necrosis (osteonecrosis) of the femoral head
- Hip dysplasia requiring arthroplasty
- Post-traumatic arthritis (following hip fracture or dislocation)
- Revision of failed hip arthroplasty (where acetabular component needs replacement)
INDICATIONS
- Surgical preparation (reaming) of the acetabulum to receive a hip implant during primary or revision hip arthroplasty.
- Use in patients with a diseased, damaged, or malformed acetabulum requiring surgical intervention and prosthetic joint replacement.
- Intended for use by trained orthopedic surgeons in a sterile surgical environment.
CONTRAINDICATIONS
- Active infection (local or systemic)
- Skeletal immaturity (patients whose bone growth is not complete)
- Severe vascular insufficiency, neuromuscular, or other systemic disease that precludes safe surgery
- Inadequate bone stock or poor bone quality that cannot support an implant
- Patients with insufficient bone healing capacity (e.g., metabolic bone disease, severe osteoporosis)
STORAGE AND HANDLING
The Single-Use Acetabular Reamer should be handled with care at all times. Storage zones for surgical instruments should be away from areas of humidity and must be out of contact with UV rays and sources of electro-magnetic radiation.
Do not use the Single-Use Acetabular Reamer after the expiration date indicated on the package and label. After this date, the sterility of the instruments can no longer be assured.
WARNINGS
WARNINGS:
- Do not use if package damaged.
- Do not reuse.
- Inspect locking interface prior to activation.
- Ensure proper coupling before activation.
- Ensure adequate irrigation to minimize heat.
- Avoid excessive axial force.
CAUTION
Federal Law (USA) restricts these instruments to sale by or on the order of a physician.
LIMITED PRODUCT WARRANTY
Refer to www.osteofitllc.com/limited-warranty for limited product warranty terms and conditions.